Gestational Diabetes in Uganda and India: Design and Evaluation of Educational Films for improving Screening and Self-management (GUIDES) is a cluster randomized controlled trial to evaluate a package of interconnected educational/behavioural interventions aimed at: a) improving knowledge and skills of Gestational Diabetes Mellitus (GDM) guidelines and skills of health providers; b) raising awareness of importance of GDM screening among pregnant women and their families; and c) improving confidence and skills in self-management among those diagnosed with GDM. The interventions will be delivered through the medium of film as they are low-cost and scalable, and are particularly suitable for people who are not very literate.
The research is ongoing in India (Bengaluru) and Uganda (Mpigi, Wakiso and Masaka). Following a careful contextual analysis, we have worked with our partner Medical Aid Films to develop a set of educational films for pregnant women and healthcare staff. The effectiveness of this film-based intervention is being evaluated in two independent cluster randomized trials, involving ~10,000 pregnant women across 30 maternity units in Uganda and 30 maternity units in India. Our study outcomes are: a) detection of GDM at 32 weeks of; b) glycaemic control (fasting glucose) in women with GDM at ~34 weeks of pregnancy; and c) adverse perinatal outcomes.
GUIDES is being led by the London School of Hygiene and Tropical Medicine (UK), the Public Health Foundation of India, and the MRC/UVRI and LSHTM Uganda Research Unit.
The study sponsor is the London School of Hygiene and Tropical Medicine.
GUIDES is funded by the Newton Fund Programme via the Medical Research Council and UK Research and Innovation (ref: MR/R021392), and by the Department of Biotechnology, Government of India (ref: BT/IN/DBT-MRC/DIFD/GRB/17/2018-19).
Trial registration:https://clinicaltrials.gov/ct2/show/NCT03937050
India trial registration: CTRI number: CTRI/2020/02/023605 [Registered on: 26/02/2020]
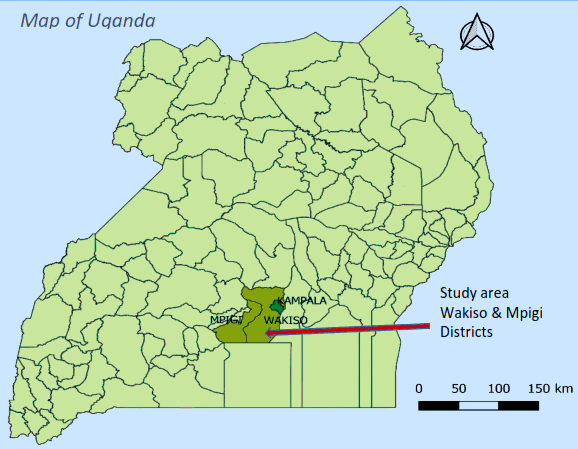
India Study Sites
Uganda Study Sites